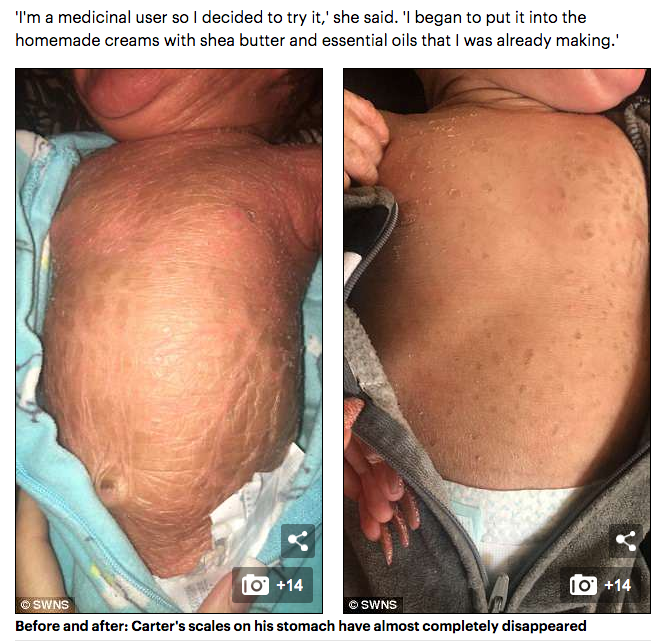
The discovery of the endocannabinoid system (ECS) is the single most important
scientific medical discovery since the recognition of sterile surgical technique. As our
knowledge expands, we are coming to realize that the ECS is a master control system of
virtually all physiology. The total effect of the ECS is to regulate homeostasis and prevent
disease and aging.
The more we learn, the more we realize that we are in the infancy of this scientific field of study. The ECS is a control system which involves tissue receptor proteins, cellular communication and control, molecular anatomy and the scavenging of oxygen free radicals. This new field of science will change medicine forever and prove cannabis the gold standard for many disease processes. Its effect on scavenging oxygen free radicals is applicable to all disease processes and this is why it has such wide medical application and is considered a cure-all by many.
The discovery of the ECS will replace the current medical system of managing and
treating disease. Instead of management of symptoms after disease has occurred, we will prevent disease and cancer by manipulation of the ECS. Research and education of medical students involving the ECS is being intentionally restricted by politics. No justification can be made for the restriction of the scientific study of cannabis and the endocannabinoid system. What is the danger of providing government-grown
and tested cannabis to researchers?
Diversion of research cannabis for non-scientific or recreational purposes does not seem to be a serious threat to national security.
The issue of cannabis abuse and dependency remains quite controversial. A cannabis dependency syndrome has been posited (Budney et al., 2004), with an oft quoted figure of 9% of ever cannabis users becoming dependent at some point. In the USA, at least, these figures, which apply to “recreational” usage must be tempered by the fact that greater than 50% of patients admitted to substance abuse treatment programs are there by legal mandate as an alternative to prosecution or incarceration, and not always because of an actual addiction to cannabis. Other authorities opine that cannabis has a DAL lower than that of other legal and illicit agents (Hilts, 1994; Roques, 1998; Nutt et al., 2007). The relative addictive potential of a drug is ascertained by judging its attendant intoxication, reinforcement, tolerance, withdrawal and dependency. DAL requires additional determination of public health and legal data on its degree of abuse and diversion. The advent of the Internet has revolutionized promulgation of drug information to any inquisitive potential consumer.
Herbal cannabis is scheduled in international and national categories that generally designate it as addictive or dangerous, having severe abuse potential, and lacking any recognized medical utility. In contrast, Marinol®, a synthetic form of THC has been down-scheduled in countries where it is an approved pharmaceutical, to a category denoting a lesser potential for abuse or lower dependency risk, after documentation showed rare abuse or diversion to the black market (Calhoun et al., 1998). This precedent is one that could potentially be repeated with cannabis-based medicines once their safety and appropriate DAL risk is demonstrated.
Tolerance is quickly established to various manifestations of cannabinoid intoxication: tachycardia, hypothermia, orthostatic hypotension, dry mouth, ocular injection, intraocular pressure decreases, etc. (Jones et al., 1976). In over 15,000 patient-years of experience, no dose tolerance to nabiximols has been observed, however, while therapeutic efficacy is maintained (Wade et al., 2006; Notcutt et al., 2012; Serpell et al., 2013) In SAFEX studies in MS and peripheral neuropathic pain, nabiximols doses have been steady or reduced after months or years of administration (Serpell et al., 2013; Koehler, 2014). Symptomatic pain control is maintained with slow continued improvement in non-progressive disorders.
The existence or severity of a cannabis withdrawal syndrome remains under debate (Smith, 2002; Budney et al., 2004). In contrast to reported withdrawal sequelae in recreational users (Solowij et al., 2002), 24 subjects with MS who volunteered to discontinue nabiximols after a year or more suffered no withdrawal symptoms meeting Budney criteria. While symptoms such as pain recurred after some 7–10 days without Sativex, symptom control was rapidly re-attained upon resumption (Wade et al., 2006). Similar safety was noted in a clinical randomized withdrawal trial in spasticity of MS (Notcutt et al., 2012), wherein 36 patients previously improved on Sativex showed no withdrawal symptoms of significance. Additionally, in a study of 136 MS patients taking Sativex for a mean of 334 days, sudden cessation, no withdrawal effects of associated adverse events were reported (Serpell et al., 2013).
While herbal cannabis has lowest overall dependency risk of commonly abused drugs (Hilts, 1994; Roques, 1998; Nutt et al., 2007), that of nabiximols is apparently lower yet, due to slower peak compared to smoking, low doses required for therapeutic efficacy, virtual absence of intoxication in normal usage, and freedom from withdrawal sequelae even after chronic administration. Finally, no known abuse or diversion incidents with nabiximols were reported (as of March 2013). Formal DAL studies with nabiximols have demonstrated its drug abuse potential to be equal to or less than that of Marinol, which is Schedule III in the USA (Schoedel et al., 2011).
The situation is particularly puzzling for cannabidiol, which seems to be a victim of guilt by association, in that it was placed in Schedule I of the US Controlled Substances Act of 1970 along with cannabis and THC as a placeholder (United States Commission on Marihuana and Drug Abuse, 1972), and has remained there ever since, in spite of meeting no criteria for intoxication, reinforcement, tolerance, withdrawal, or dependency.
Since cannabis can stay in your system for weeks, it’s been demonized, wrongfully accused of being addictive because it does not stimulate that reward centre.
This is interesting, I guess that’s why patients who rely heavily on cannabis, don’t really get high. I wonder if they can fix that? 🙂
Science Daily published – Cannabis: Non-addictive pathway to pain relief? Medicinal properties of cannabis examined
Ingram and colleagues report the treatment of chronic pain has challenged the medical system, with medications that are ineffective or create serious side effects: “However, emerging data indicate that drugs that target the endocannabinoid system might produce analgesia with fewer side effects compared with opioids.”
The body’s endocannabinoid system comprises receptors, endocannabinoid molecules and enzymes that make and degrade the endocannabinoids located in the brain and throughout the central and peripheral nervous system. The research team focused on two cannabinoid receptors, known as CB1 and CB2, in the rostral ventromedial medulla — a group of neurons located in the brainstem known to modulate pain. The study is the first to examine CB1 and CB2 receptor function at the membrane level in late adolescent and adult neurons.
The researchers observed that chronic inflammatory pain increased activity of CB2 receptors and decreased CB1 activity. Cannabis activates both CB1 and CB2 receptors equally. The study suggests that selective activation of CB2 receptors contributes to the medicinal benefit of cannabis while minimizing the propensity of the other cannabinoid receptor, CB1, to induce tolerance and withdrawal. Ingram said the next phase of the research will further explore this area of brain circuitry, which ultimately could lead to the development of a new class of pain medications.
Co-authors include lead author Ming-Hua Li, Ph.D., and Katherine L. Suchland, both with the Department of Neurological Surgery, OHSU School of Medicine.
The study was funded by grants from the National Institutes of Health (DA035316 and R56NS093894) and American Heart Association (13SDG14590005, MH.L.).
Cannabinoids as Therapeutics , Edited by R. Mechoulam , ISBN 3-7643-7055-6 Birkhäuser Verlag, Basel – Boston – Berlin